- Share
- Share on Facebook
- Share on LinkedIn
Seminar
On March 23, 2026
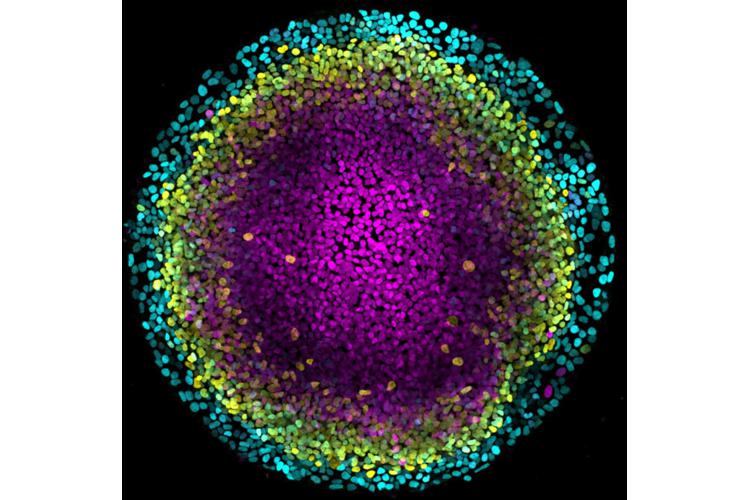
Chloé Roffay (IMP Vienna, Austria)
Extraembryonic tissues provide key molecular signals and mechanical support to the growing embryo. For instance, the extraembryonic amnion, which forms a fluid-filled sac surrounding the embryo, was recently shown to trigger germ layer specification during gastrulation, by secreting BMP ligands. Despite the key roles of extraembryonic tissues in embryo development, little is still known regarding their molecular and biophysical programs, particularly in human. Using a 2D stem cell-based model of human gastrulation, termed gastruloid discs, we found that amnion cells undergo a sharp columnar-to-squamous transition concomitantly with fate specification. Via biophysical modelling, direct force measurements, pharmacological and genetic perturbations, we showed that this morphogenetic transition is amnion-intrinsic and it is driven by active wetting. Molecularly, active wetting is implemented via a rewiring of cytoskeleton composition, from actomyosin to keratin-based cytoskeletal networks. Strikingly, blocking shape changes at the colony edge results both in defective cellular states in the amnion and impaired gastruloid disc morphogenesis within the embryonic compartment. Together, our findings establish that a cytoskeletal rewiring couples fate specification to tissue architecture in the human amnion and suggest an unexpectedly active mechanical role for extraembryonic tissues in shaping the embryo proper.
Contact: Thomas Boudou
Date
11:00
Localisation
LIPhy, salle de conférence
- Share
- Share on Facebook
- Share on LinkedIn