- Imprimer
- Partager
- Partager sur Facebook
- Partager sur LinkedIn
Séminaire
Le 3 avril 2023
Charlotte Aumeier (Université de Genève)
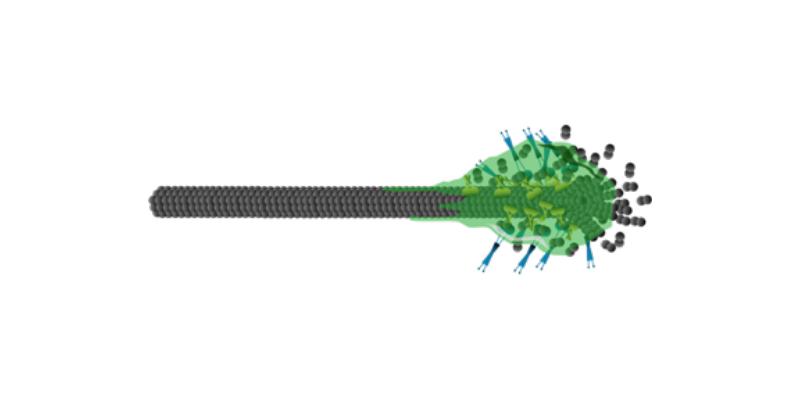
Regulation of microtubule dynamics is essential for diverse cellular functions, and proteins that bind to dynamic microtubule ends can regulate network dynamics. Here we show that two conserved microtubule end-binding proteins, CLIP-170 and EB3, undergo phase separation and form dense liquid-networks. When CLIP-170 and EB3 act together the multivalency of the network increases, which synergistically increases the amount of protein in the dense phase. In vitro and in cells these liquid networks can condense tubulin. In vitro in the presence of microtubules, EB3/CLIP-170 phase separation can co-condense tubulin all along the microtubule. At this condition microtubule growth speed increases up to two-fold and depolymerization events are strongly reduced, compared to conditions with phase separation deficient networks. Our data show that phase separated EB3/CLIP-170 networks impact microtubule growth dynamics beyond direct protein-microtubule interactions.
Date
14:00
Localisation
LIPhy, salle de conférence
- Imprimer
- Partager
- Partager sur Facebook
- Partager sur LinkedIn